_________________________________________________________________________________________
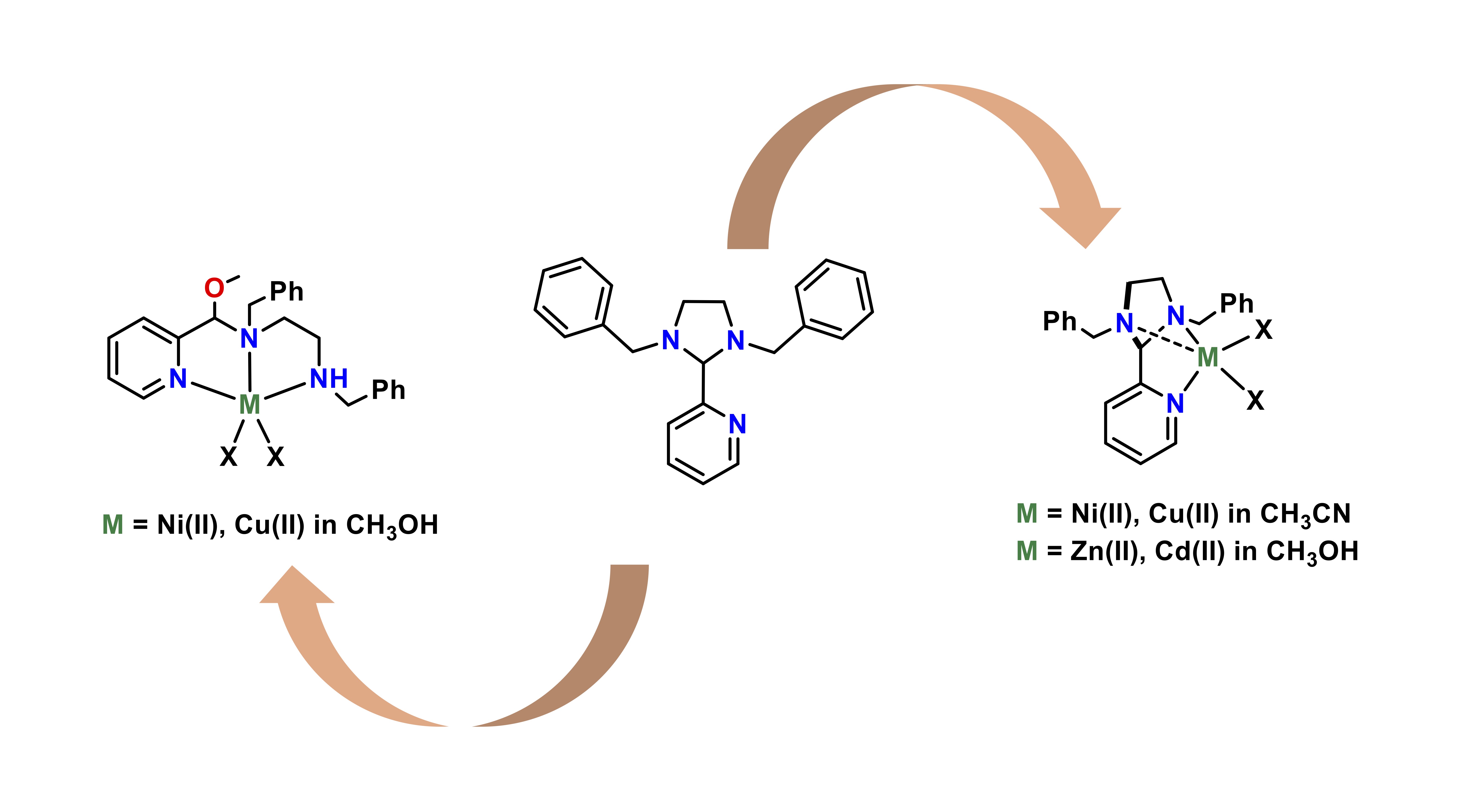
Metal and Solvent–Dependent Formation of Imidazolidine or Hemiaminal Ether Complexes
S. Ghosh, R. J. Butcher, R. Angamuthu*
_________________________________________________________________________________________
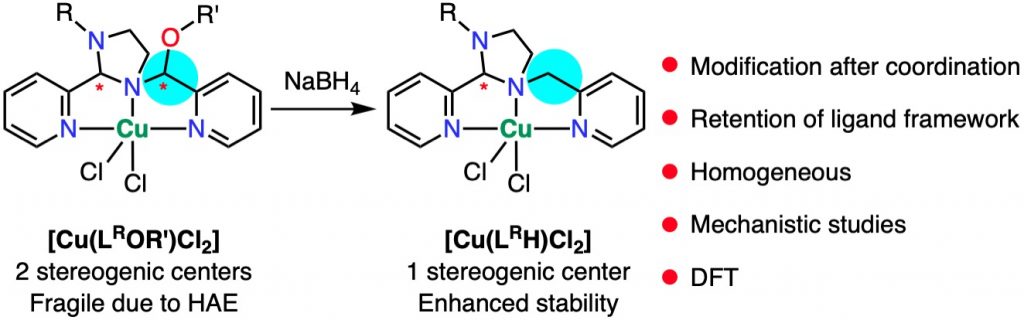
Late-stage Ligand Modification After Coordination Strengthens Stereoselectively Self-Assembled Hemiaminal Ether Complexes
S. Raje, A. Koner, S. Ghosh, R. J. Butcher, L. Mathivathanan, M. Sundararajan, R. Angamuthu*
________________________________________________________________________________________
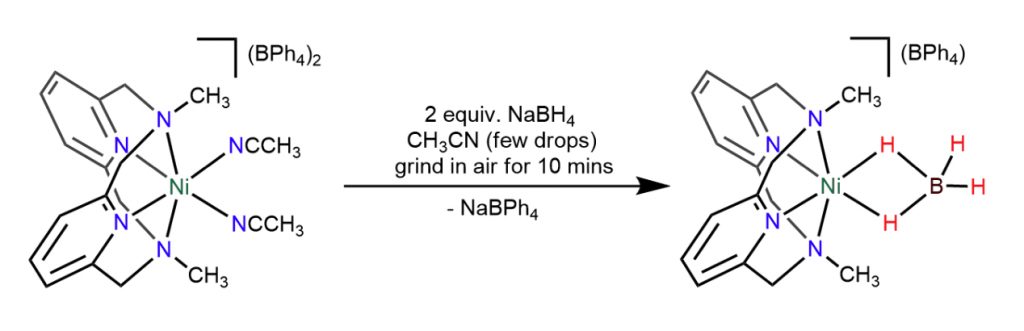
Mechanochemical Synthesis and Reactivity of a Stable Nickel Borohydride
S. Raje,* K. Mani, S. Dinesh, A. Yadav, M. Chahal, R. J. Butcher, and R. Angamuthu*
_________________________________________________________________________________________
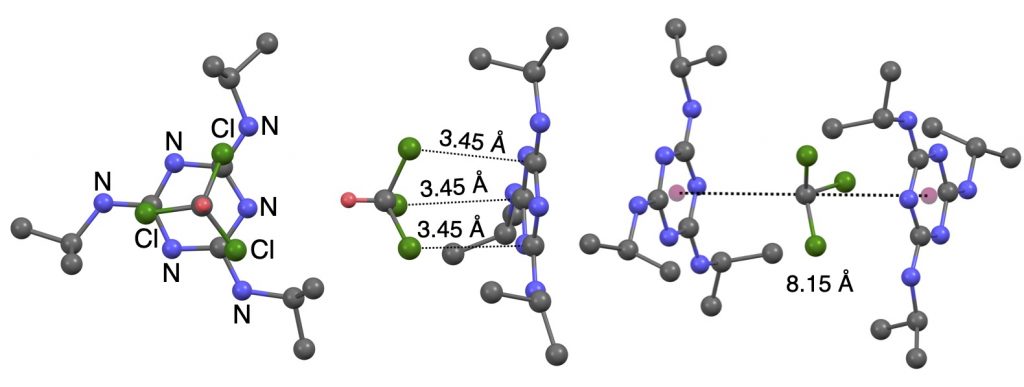
Triazine-based eccentric Piedfort units towards a single source hydrogen-bonded network.
S. Mehrotra, S. Raje, A. K. Jain, R. J. Butcher and R. Angamuthu*
_________________________________________________________________________________________
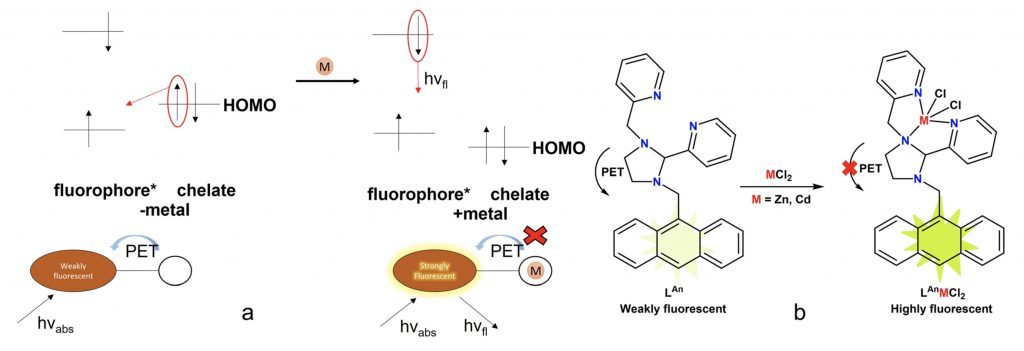
Investigating the photosensitivity of koneramines for cell imaging and therapeutic applications.
S. Ghosh,* A. Akhir, D. Saxena, S. Singh, S. Sivakumar, S. Chopra* and R. Angamuthu*
_________________________________________________________________________________________
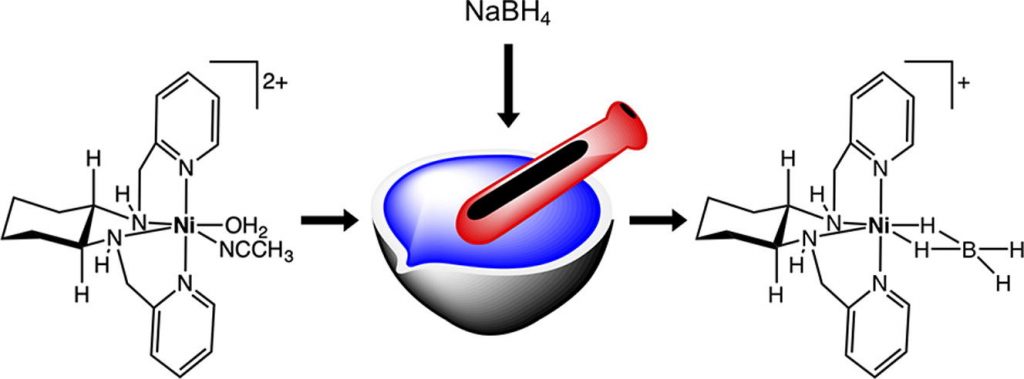
Air-stable nickel(II) borohydrides as prohydrides: Reactions with halocarbons and aerial carbon dioxide
K. Mani, P. Pal, K. Anandababu, S. Raje,* R. Angamuthu*
_________________________________________________________________________________________
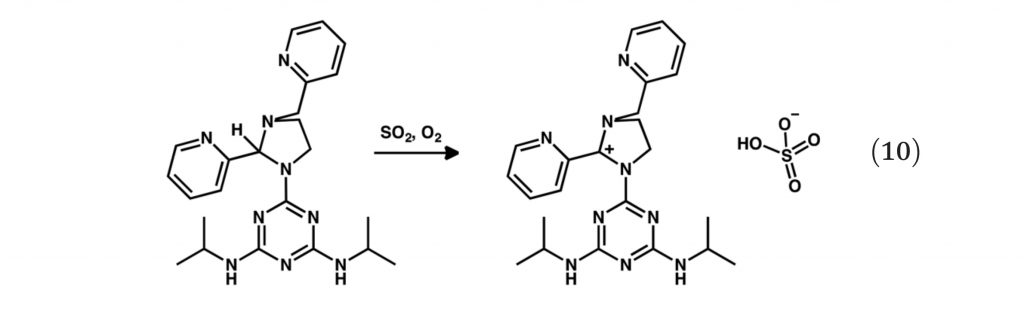
Binding enabled catalytic activation of SO2 by copper koneramine complexes under ambient conditions.
M. Chahal, S. Raje,* G. Kotana and R. Angamuthu*
_________________________________________________________________________________________
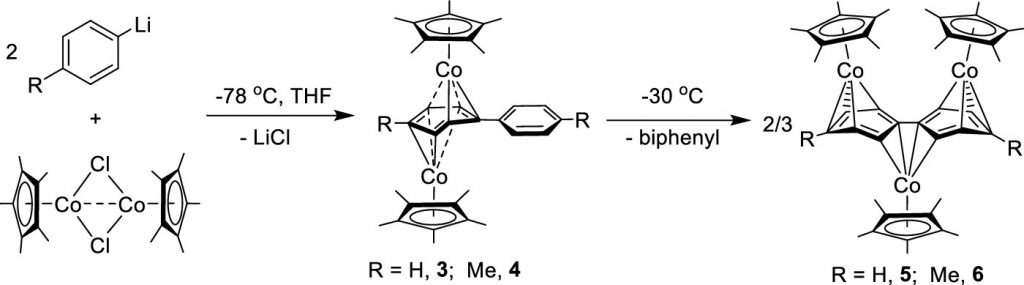
Reductive Coupling of Bridging Diaryl Ligands in Half-Sandwich Cobalt(II) Dimers: Revisiting Triple-Decker Cobalt(I) Complexes.
S. Zhang, Y. Song, X. He, R. Angamuthu,* C.-H. Tung, and W. Wang*
_________________________________________________________________________________________
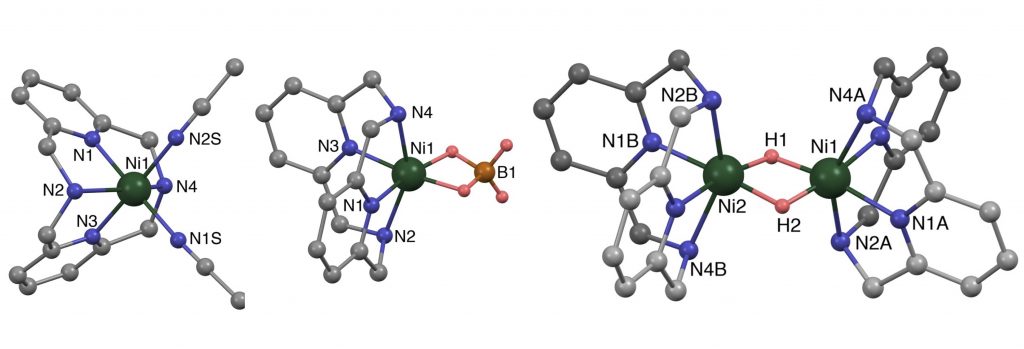
Solvent-Free Synthesis and Reactivity of Nickel(II) Borohydride and Nickel(II) Hydride.
S. Raje* and R. Angamuthu*
_________________________________________________________________________________________
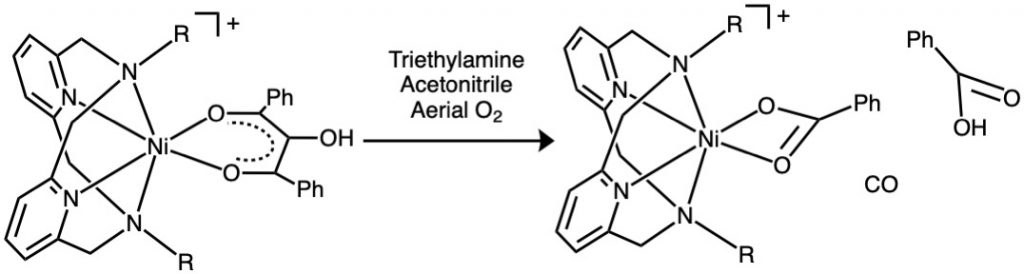
Bioinspired Oxidative Cleavage of Aliphatic C-C Bond Utilizing Aerial Oxygen by Nickel Acireductone Dioxygenase Mimics.
S. Raje, K. Mani, P. Kandasamy, R. J. Butcher and R. Angamuthu*
_________________________________________________________________________________________
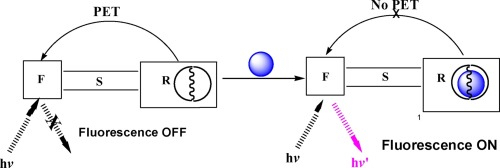
Cryptand-based fluorescence signalling systems for transition, inner-transition, and main group metal ions: Modulation of the PET process through the attachment of electron-withdrawing and electron-donating groups
D. Bharadwaj, R. Angamuthu*
_________________________________________________________________________________________
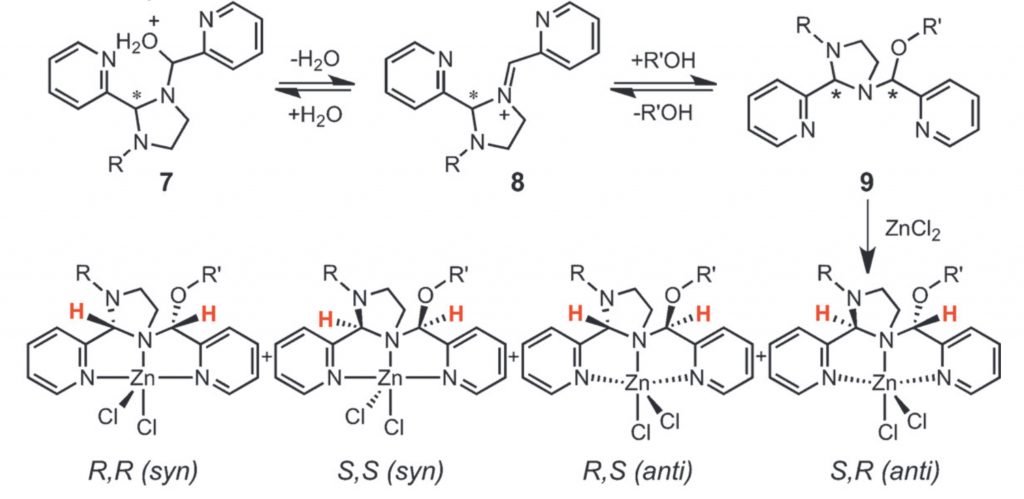
Mechanism of Evolution of Koneramine Complexes from One‐Pot Reactions: Snapshots of Intermediates Offer Facile Routes to New Dipicolylamines.
S. Raje, N. Mondivagu, M. Chahal, R. J. Butcher, R. Angamuthu*
_________________________________________________________________________________________
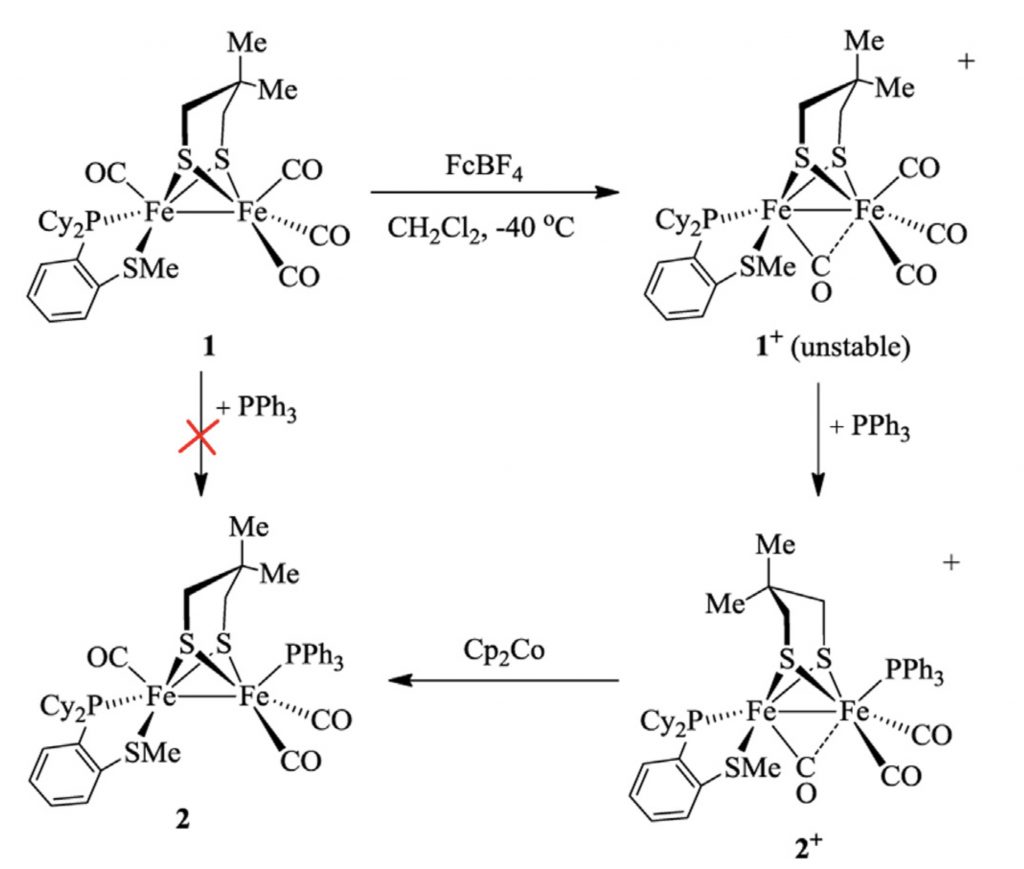
Synthetic [FeFe]-H2ase models bearing phosphino thioether chelating ligands
Y. Zhao, X. Yu, H. Hu, X. Hu, S. Raje, R. Angamuthu, C-H. Tung, W. Wang*
_________________________________________________________________________________________
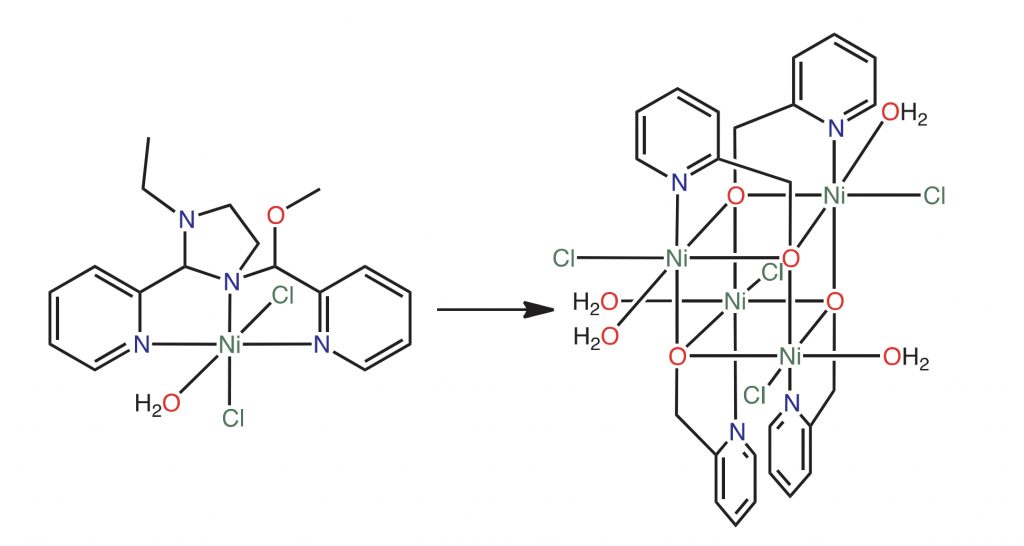
Tetranuclear nickel cubane cluster formed by the hydrolysis of nickel koneramine complex
S. Raje,* R. Angamuthu
_________________________________________________________________________________________
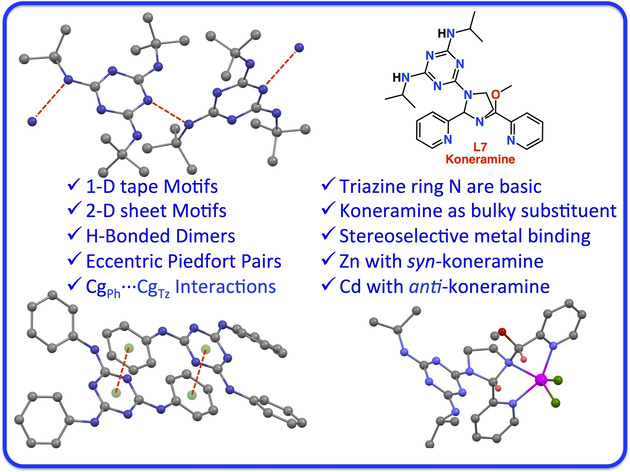
A Quest towards Eccentric Piedfort Pairs
S. Mehrotra, S. Raje, A. K. Jain, A. Jain, P. Kandasamy, R. J. Butcher, and R. Angamuthu*
_________________________________________________________________________________________
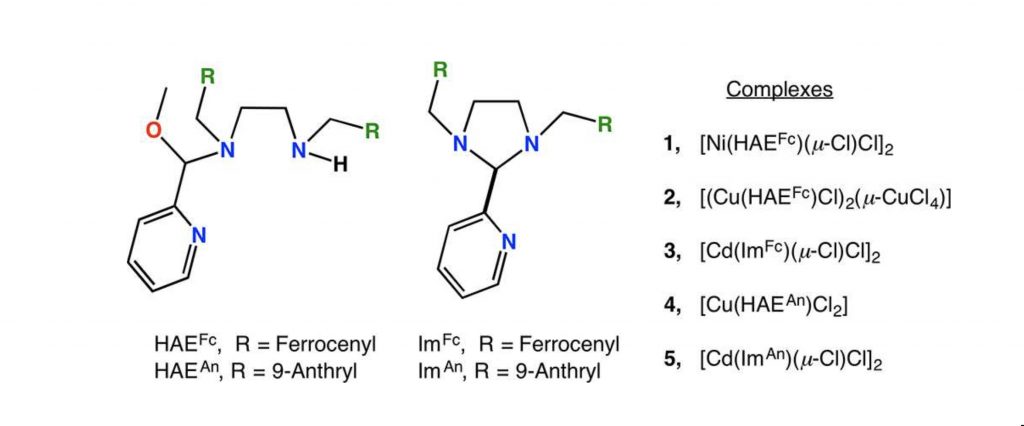
Metal Dependent Formation of Imidazolidine or Hemiaminal Ether Complexes from Multicomponent One-pot Reactions
M. Chahal, K. Mani, C. S. Lodhi, R. J. Butcher, S. Raje,* R. Angamuthu*
_________________________________________________________________________________________
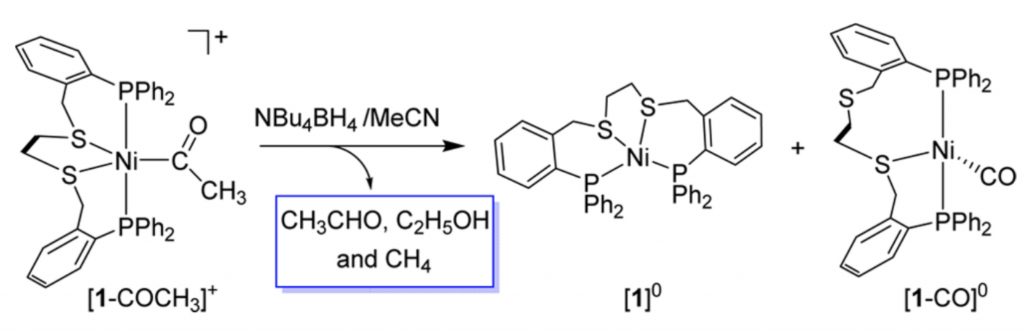
Nickel-Mediated Stepwise Transformation of CO to Acetaldehyde and Ethanol.
A. Zhang, S. Raje, J. Liu, X. Li, R. Angamuthu, C.-H. Tung, W. Wang*
_________________________________________________________________________________________
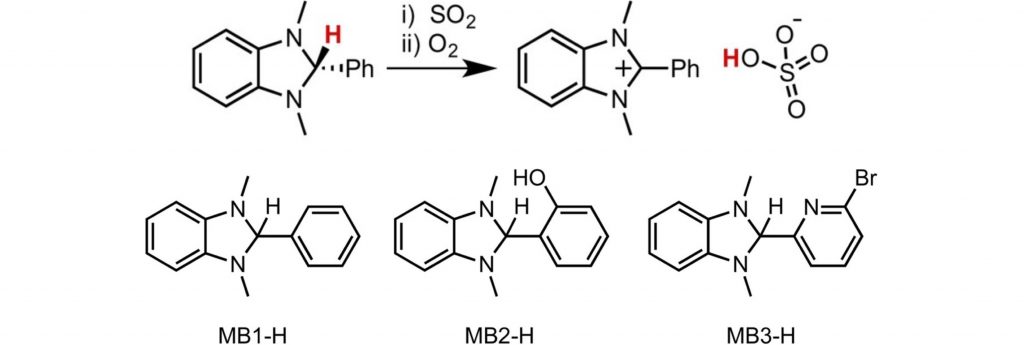
Benzimidazolines Convert Sulfur Dioxide to Bisulfate at Room Temperature and Atmospheric Pressure Utilizing Aerial Oxygen.
S. Mehrotra, S. Raje, A. K. Jain, R. Angamuthu*
_________________________________________________________________________________________
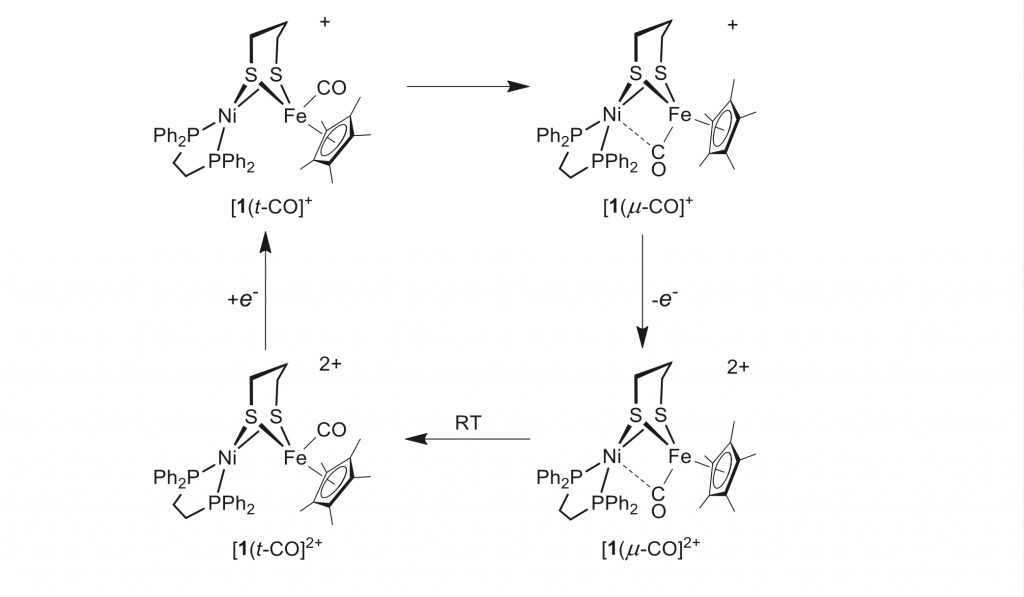
Synthetic [NiFe] models with a fluxional CO ligand.
X. Chu, X. Yu, S. Raje, R. Angamuthu, J. Ma, C.-H. Tung, W. Wang*
_________________________________________________________________________________________
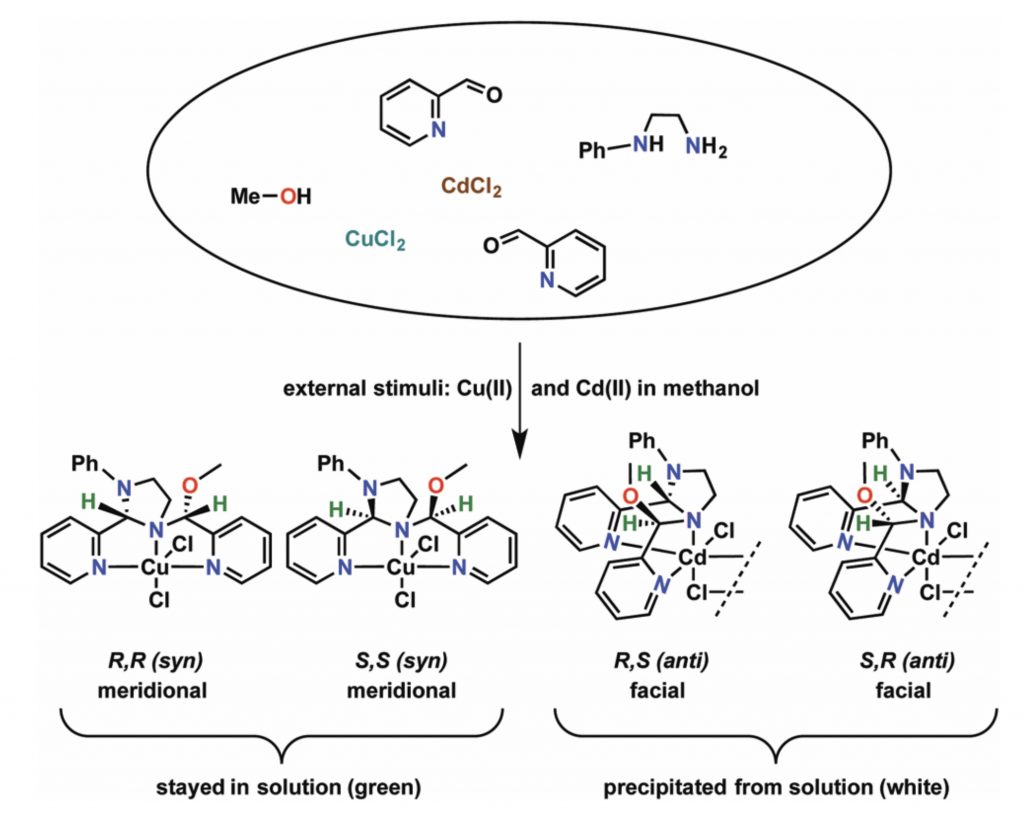
Metal Ions as External Stimuli in Stereoselective Self-Sorting of Koneramines and Thiokoneramines
S. Raje, K. Mani, M. M. Parsutkar and R. Angamuthu*
_________________________________________________________________________________________
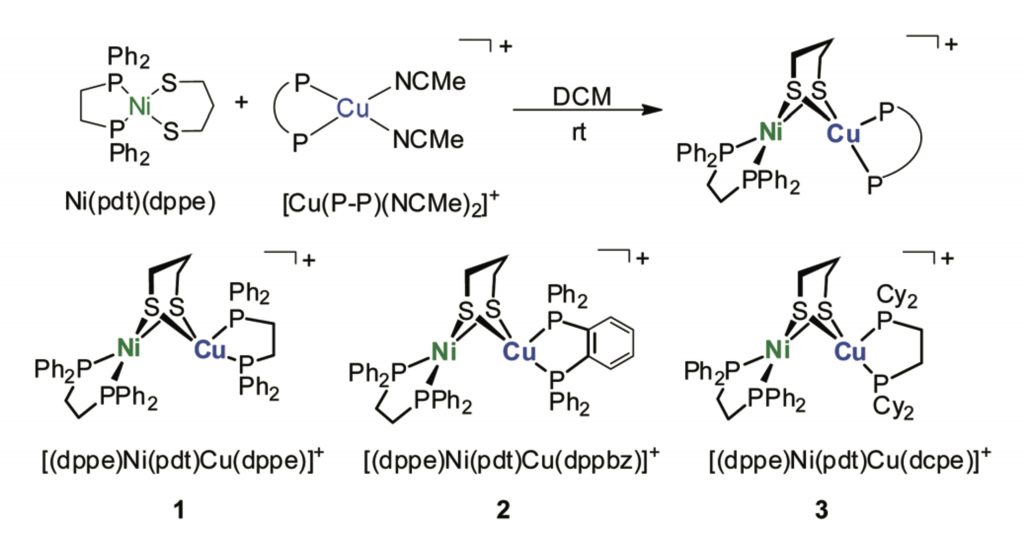
Heteronuclear assembly of Ni–Cu dithiolato complexes: synthesis, structures, and reactivity studies
X. Chu, X. Xu, H. Su, S. Raje, R. Angamuthu, C-H. Tung and W. Wang*
_________________________________________________________________________________________
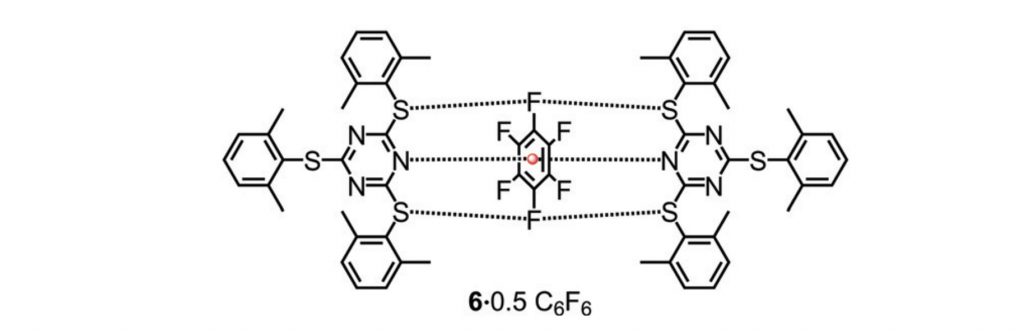
Janus head type lone pair–π–lone pair and S⋯F⋯S interactions in retaining hexafluorobenzene
S. Mehrotra and R. Angamuthu*
_________________________________________________________________________________________
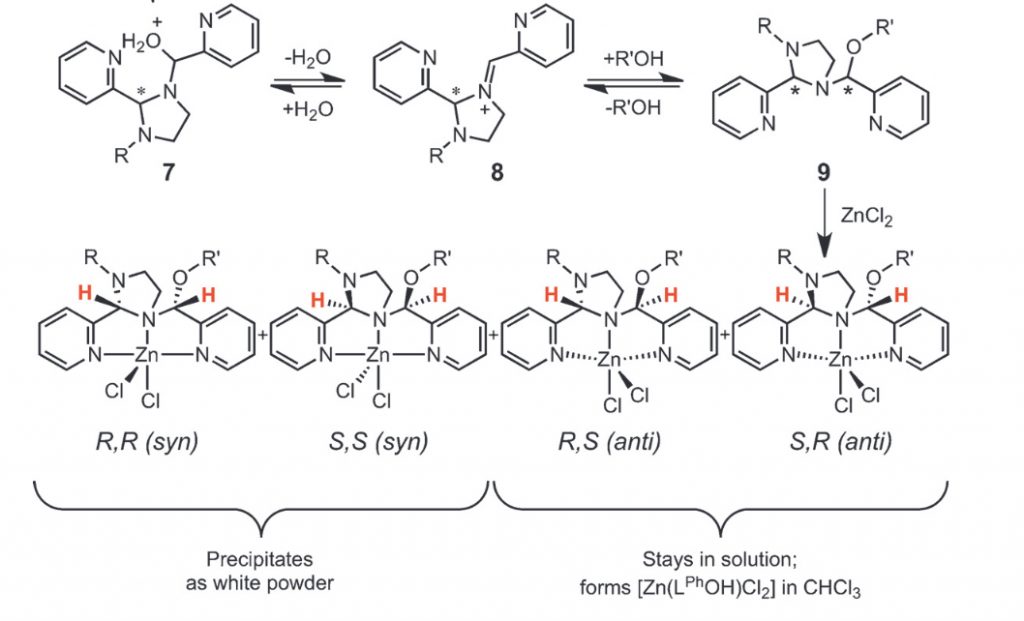
Multicomponent One-pot Reactions Towards the Synthesis of Stereoisomers of Dipicolylamine Complexes.
S. Raje, S. Gurusamy, A. Koner, S. Mehrotra, S. J. Jennifer, P. G. Vasudev, R. J. Butcher, R. Angamuthu*
_________________________________________________________________________________________
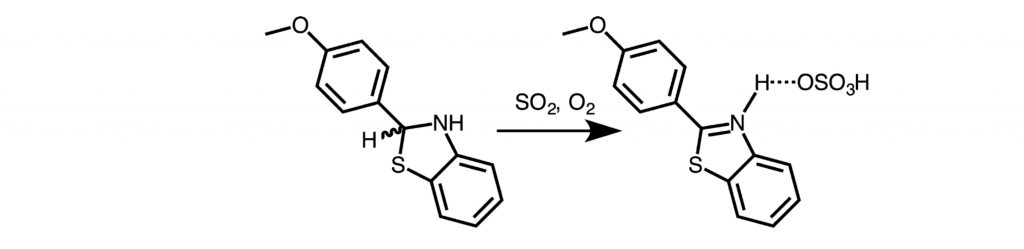
Benzothiazoline Converts SO2 to Sulfuric Acid en Route to Benzothiazole.
S. Mehrotra, R. J. Butcher, R. Angamuthu*
********************************************************************************************************************************************************
PDF (University of Illinois at Urbana-Champaign; Prof. Thomas B. Rauchfuss)
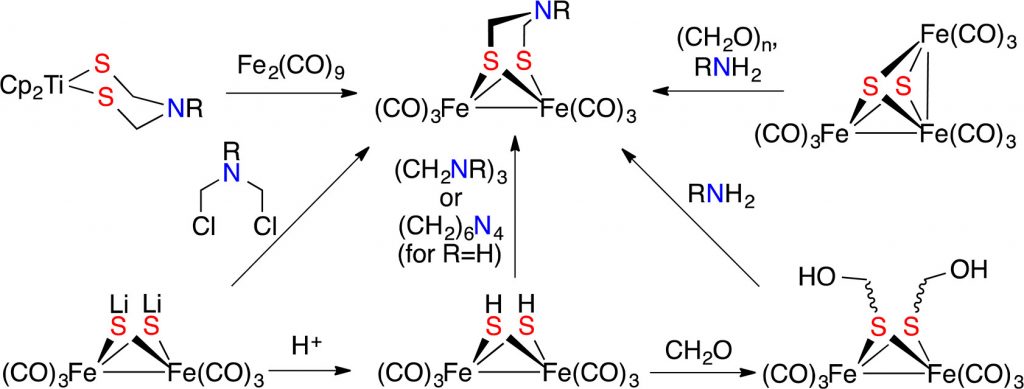
R. Angamuthu, C.-S. Chen, T. R. Cochrane, D. L. Gray, D. Schilter, O. A. Ulloa, T. B. Rauchfuss*
N-Substituted Derivatives of the Azadithiolate Cofactor from the [FeFe] Hydrogenases: Stability and Complexation.
_________________________________________________________________________________________
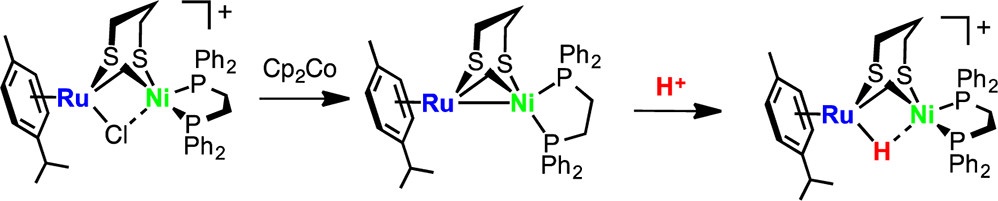
G. M. Chambers, R. Angamuthu, D. L. Gray, T. B. Rauchfuss*
Organo Ruthenium–Nickel Dithiolates with Redox-Responsive Nickel Sites.
_________________________________________________________________________________________
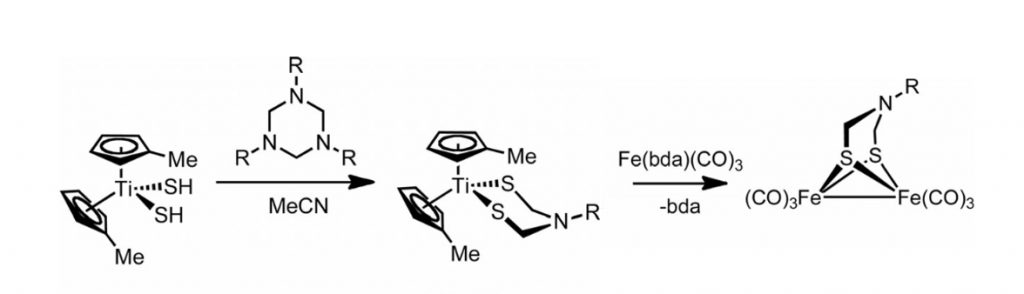
R. Angamuthu, M. E. Carroll, M. Ramesh, T. B. Rauchfuss*
A New Route to Azadithiolato Complexes.
_________________________________________________________________________________________
********************************************************************************************************************************************************
PhD (Leiden University, The Netherlands; Profs Jan Reedijk and Elisabeth Bouwman)
_________________________________________________________________________________________
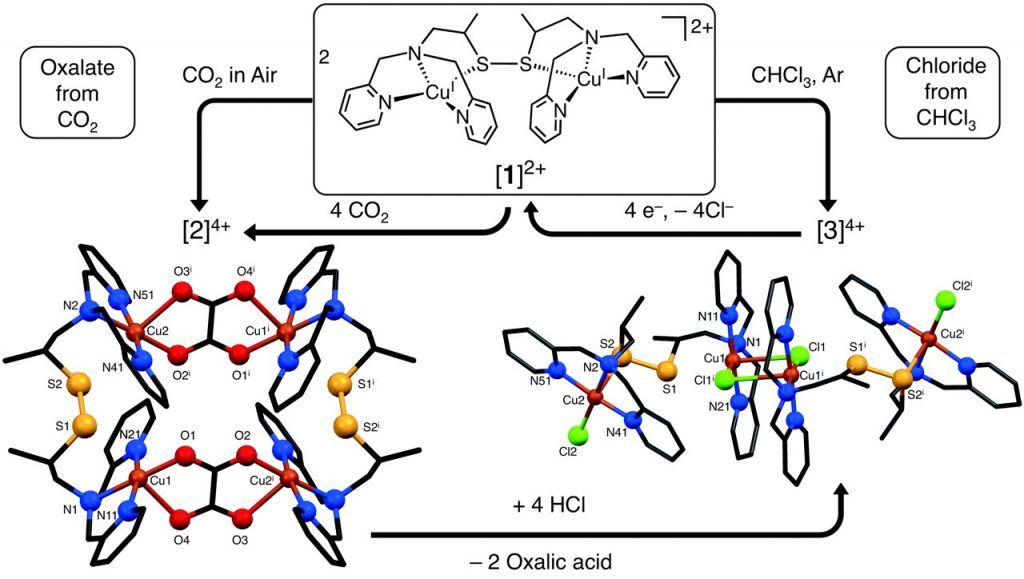
R. Angamuthu, P. Byers, M. Lutz, A. L. Spek, E. Bouwman*
Electrocatalytic carbon dioxide Conversion to Oxalate by a Copper Complex.
_________________________________________________________________________________________
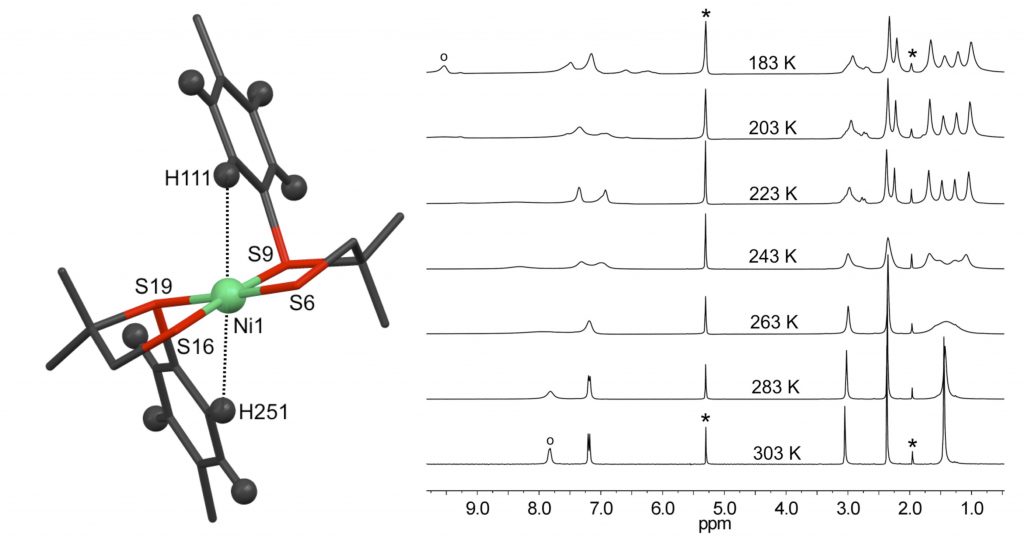
R. Angamuthu, L. L. Gelauff, M. A. Siegler, A. L. Spek, E. Bouwman*
A molecular cage of nickel(II) and copper(I): a [{Ni(L)2}2(CuI)6] cluster resembling the active site of nickel-containing enzymes.
_________________________________________________________________________________________
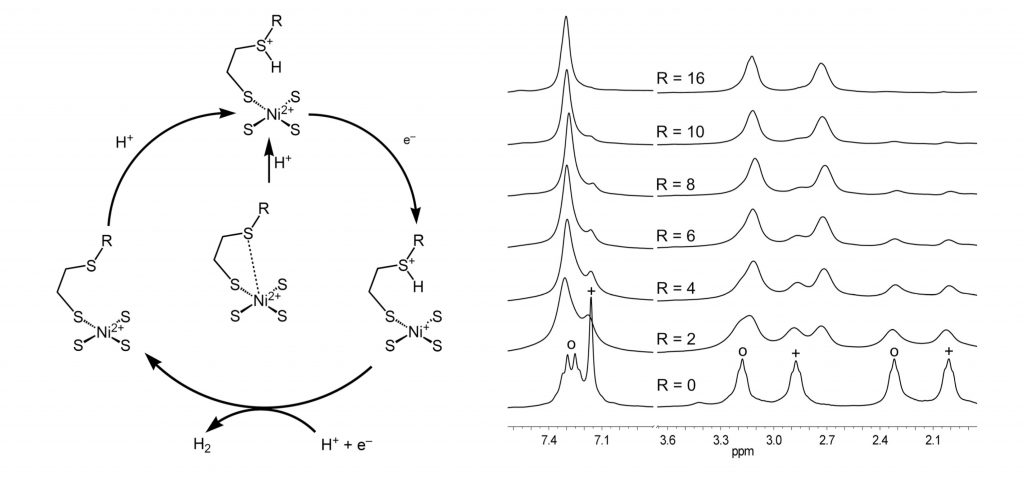
R. Angamuthu, E. Bouwman*
Reduction of protons assisted by a hexanuclear nickel thiolate metallacrown: protonation and electrocatalytic dihydrogen evolution.
_________________________________________________________________________________________
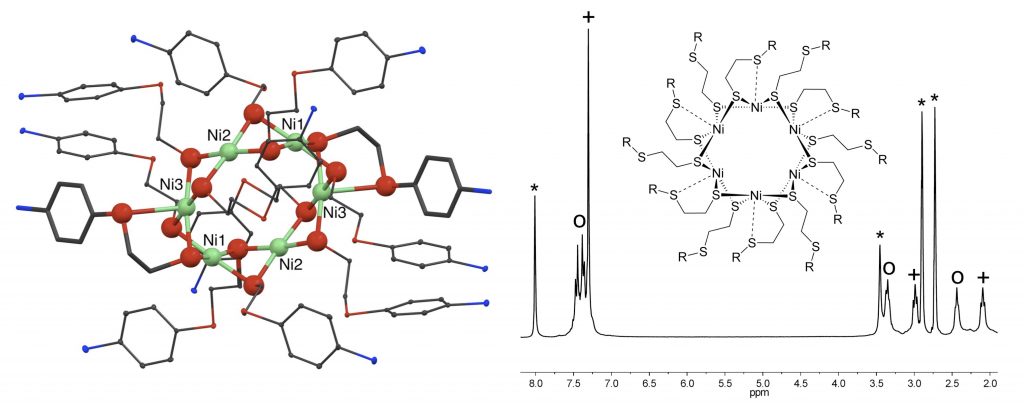
R. Angamuthu, H. Kooijman, M. Lutz, A. L. Spek, E. Bouwman*
Hexanuclear [Ni6L12] metallacrown framework consisting of NiS4 square-planar and NiS5 square-pyramidal building blocks.
_________________________________________________________________________________________
********************************************************************************************************************************************************
MSc (Bharathidasan University, Tiruchirappalli; Prof. Mallayan Palaniandavar)
_________________________________________________________________________________________
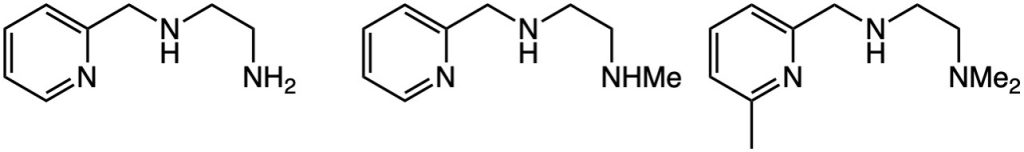
R. Angamuthu, R. Venugopal, U. M. Palanisamy, B. Ramalingam, C. A. Kilner, M. A. Halcrow, P. Mallayan*
Copper(II) complexes of tridentate pyridylmethylethylenediamines: Role of ligand steric hindrance on DNA binding and cleavage.
_________________________________________________________________________________________
********************************************************************************************************************************************************
